Should we pay to play? Charging for New Zealand’s landscape
Posted: May 1, 2018 Filed under: 2018, ERES525, Research Essay | Tags: conservation, DOC, ecotourism, New Zealand Leave a comment
Mt Taranaki – Harley Betts
By Daniel Papworth
New Zealand (NZ) has some of the most beautiful landscape in the entire world. However some areas are getting overrun by tourists. Over the past year 3.8 million people visited NZ’s shores[1]. Over half of them explored at least one national park or protected area[2].
NZ’s National parks system aims to preserve the country’s intrinsic worth for the enjoyment of the public. Park land contains “scenery of such distinctive quality, ecological systems, or natural features so beautiful, unique, or scientifically important that their preservation is in the national interest”[3]. The Department of Conservation (DOC) are charged with managing our parks, however they are seriously underfunded having around $20 a hectare of land they manage[4]. As a consequence from over use and lack of resources the beauty of these places may be at risk. Combined with climate change stress in these environments is mounting[5, 6]. Currently under the National Parks Act (1980) DOC is unable to charge for access. Regulation of the number of people visiting these area’s needs to be implemented to conserve them and their beauty.
Travelling abroad, you will almost always be charged to enter a national park. Chris Roberts, CEO of Tourism Aotearoa, says that only half a dozen sites in NZ are in serious need of number management[7]. Shouldn’t we then charge tourists to see these high use sites? Money spent on entry can be directed straight back into the maintenance of these tourist hotspots. The number of people visiting at one time can be monitored and regulated if needed. Another option is a ‘conservation tax’ or ‘nature levy’. When entering NZ international visitors could pay a small sum to be given to DOC for the upkeep of these high-use areas. If 3.8 million people visit a year and each is charged $20 that comes to seventy-six million dollars, nearly a quarter of DOC’s yearly budget. The total cost to visit NZ would then be $50, still cheaper than Australia’s $58-$85 border fee[8].
So why haven’t we used any of these methods yet? The problem is relatively new. Visitor numbers have increased from 2.6-3.8 million in the last five years[9]. In recent years DOC’s budget has reduced, making their job of maintaining these areas and conserving NZ’s endangered species all the more difficult. There is also likely hesitance to the investment for infrastructure. Buildings will need to be erected, people would have to be employed to process visitors. The difficulty of putting infrastructure in place could be avoided with the border tax option. However not every visitor is going to visit a national park, and some may visit multiple. Perhaps this information needs to be collected during the visa process. Or even just a free online booking system, purely for regulation purposes. Personally I feel that New Zealanders take great pride in their natural heritage. We have a certain expectation that these sort of things should free and easy to access. It feels less intrepid when you have to wait in a que to see it, less raw.
Over Easter weekend I had the pleasure of hiking the Northern Circuit in the central North Island. The locals say they cannot believe so many people still come to walk the Tongariro Alpine Crossing. Often commenting that so many come to do it they’re surprised everyone hasn’t already done it. That Easter Sunday over 3000 people walked the crossing. All those people winding their way up the mountain, using the toilets, wearing the track, stressing the fragile alpine environment[10]. Surely people will pay a small fee to experience these kind of areas. They didn’t pay to fly all the way around the globe to turn around at the gate because of a $10 entry fee. Visitors are great for the NZ economy, and these areas should be shared, but the need for number management is increasing.
So should we pay to play? Absolutely. More regulation is needed in these hot-spots for their longevity. Reducing the stress on these areas by having a charge is an opportunity we would be silly not to do. Let’s use tourism to help fund conservation. Let’s keep NZ beautiful.
References
- Statistics New Zealand (2018). Retrieved from https://www.stats.govt.nz/news/annual-visitor-arrivals-up-more-than-1-2-million-in-five-years
- Department of Conservation (2017). International Visitors Survey. Retrieved from http://www.doc.govt.nz/2017-annual-report-factsheets/?report=IVS_exp_by_NPk__2017_08_28_DOC_factsheet_template
- National Parks Act, No 66 (1980). Retrieved from http://www.legislation.govt.nz/act/public/1980/0066/latest/whole.html
- DOC is in desperate need of more funding (May 2017), Newshub. Retrieved from http://www.newshub.co.nz/home/new-zealand/2017/05/jesse-mulligan-doc-in-dire-need-of-more-funding.html
- Scott, D. (2003, April). Climate change and tourism in the mountain regions of North America. In 1st International Conference on Climate Change and Tourism (pp. 9-11).
- Moreno, A., & Becken, S. (2009). A climate change vulnerability assessment methodology for coastal tourism. Journal of Sustainable Tourism, 17(4), 473-488.
- Visitors will keep coming if national park fees introduced (February 2017), Newshub. Retrieved from http://www.newshub.co.nz/home/new-zealand/2017/02/visitors-will-keep-coming-if-national-parks-fees-introduced-industry.html
- New Zealand Green Party (2017). Tourism Levy Policy. Retrieved from https://www.greens.org.nz/policy/cleaner-environment/taonga-levy
- Statistics New Zealand (2018). Retrieved from https://www.stats.govt.nz/news/annual-visitor-arrivals-up-more-than-1-2-million-in-five-years
- Groot, R. (2003). The Tongariro National Park: Are We Loving it to Death? New Zealand Journal of Geography, 115(1), 1-13.
Confinement or Conservation? The role zoos play in the conservation movement
Posted: May 3, 2017 Filed under: 2017, ERES525, Research Essay | Tags: captive breeding, conservation, World Association of Zoos and Aquariums, zoos Leave a commentThe popularity of zoological institutions has been steadily declining in recent years, as public perception and approval of keeping animals in captivity decreases (Whitworth, 2012). This has lead to an evolution in the role of zoos, with many zoos moving away from strictly entertainment based businesses towards a more conservation focused, globally connected industry (Barongi et al., 2015). All members of the World Association of Zoos and Aquariums (WAZA) are now required to set conservation-relevant goals (Barongi et al., 2015). As anthropogenic threats to biodiversity in natural ecosystems, such as habitat destruction, climate change, invasive species, and over-exploitation of natural resources, continue to expand (Miller et al., 2004), virtually all ecosystems are undergoing catastrophic declines in their natural species. This is emphasised in Living Planet Index’s latest report, indicating that vertebrates have declined by as much as 58% between 1970 and 2012 (WWF, 2016). It is clear that it will not be possible to halt this decline without pursuing a range of conservation approaches. In this, collection-based institutions can play a significant role in the ex-situ conservation of many species worldwide (Bowkett, 2009).
For species whose habitat is severely threatened, ex-situ populations (outside of their natural habitat) can be maintained in zoos, acting as “arks” or reservoir populations (Rabb, 1994). Global captive breeding programs of such populations for reintroduction into their natural habitat have played a key role in the recovery of at least 17 species whose threat level has been reduced in North America, including the black-footed ferret (Howard et al., 2016) and Californian condor (Conde et al., 2011). Furthermore, the global network provided by the WAZA for the transfer of genetic material between zoological institutions assists in maintaining the genetic diversity of otherwise fragmented populations (Bowkett, 2009), retaining maximum heterozygosity and adaptive potential, avoiding inbreeding, and maintaining reproductive health of these populations (Howard et al., 2016) (Ivy, 2016).
Zoos provide unique opportunities for conservation-relevant research, benefitting not only captive populations but also the conservation management of natural populations and ecosystems. Zoos provide easy access to individuals and populations long-term, allowing researchers to attach significant life-history context to data and samples that would be unavailable from wild populations, due to inaccessible environments, cryptic behaviour of some species, and the possible impacts studies pose to animals in the wild (Barongi et al., 2015). Furthermore, the skills and knowledge acquired in terms of small populations management are critical for the protection of threatened populations in their natural ecosystems (Barongi et al., 2015).
Possibly the most important role zoos play in their contribution to conservation is the potential they play for the education and engagement of the public. Human lifestyle choices are driving the current declines seen in populations worldwide, and a revolution of humans’ behaviour is necessary to halt this decline (Barongi et al., 2015). While many people place an innate value on nature, others need to be convinced of the importance of conserving biodiversity. Due to urbanisation, more than 50% of the world’s population live in cities, a statistic that is likely to increase in coming years (Miller et al., 2004). Zoos provide an opportunity to engage urban populations with living organism in a way they would be unable to experience in their day-to-day lives (Rabb, 1994). In fact, more than 700million people visit WAZA affiliated zoos and aquariums yearly, giving zoos a unique opportunity to influence this large audience in pro-environmental and conservation behaviours, to bring about the attitude-shift needed to halt the worldwide decline of species seen today (Barongi et al., 2015). As such, many zoos have incorporated conservation messages in signs, presentations and campaigns situated around their facilities in order to engage visitors, and encourage their support of conservation goals (Barongi et al., 2015).
A wide range of conservation actions are required to halt the ongoing extreme rate of biodiversity decline seen throughout the world today. Here zoological institutions play an important role, providing reservoir populations and allowing for captive breeding programs, while also engaging the public in conservation projects and pro-environmental behaviours. Furthermore, they provide access to individuals for research purposes that may be otherwise unattainable from wild populations.
References
Barongi, R., Fisken, F.A., Parker, M. & Gusset, M. (eds) (2015). Committing to Conservation: the World Zoo and Aquarium Conservation Strategy. Gland, Switzerland: WAZA.
Bowkett, A.E. (2009). Recent Captive-Breeding Proposals and the Return of the Ark Concept to Global Species Conservation. Conservation Biology, Vol 23., no. 3, pp. 773-776.
Conde, D.A., Colchero, F., Jones, O.R., & Scheuerlein, A. (2011). An emerging role of zoos to conserve biodiversity. Science, Vol. 331, no. 6023, pp. 1390-1391.
Howard, J.G., Lynch, C., Santymire, R.M., Marinari, P.E. & Wildt, D.E. (2016). Recovery of gene diversity using long-term cryopreserved spermatozoa and artificial insemination in the endangered black-footed ferret. Animal Conservation, Vol. 19, no. 2, pp. 102-111.
Ivy, J.A. (2016). Ameliorating the loss of genetic diversity in captive wildlife populations. Animal Conservation, Vol. 19, no. 2, pp. 112-113.
Miller, B., Conway, W., Reading, R.P., Wemmer, C., Wildt, D., Kleiman, D., Monfort, S., Rabinowitz, A., Armstrong, B. & Hutchins, M. (2004). Evaluating the Conservation Mission of Zoos, Aquariums, Botanical Gardens and Natural History Museums. Conservation Biology, Vol. 18, no. 1, pp. 86-93.
Whitworth, A.W. (2012). An Investigation into the Determining Factors of Zoo Visitor Attendance in UK Zoos. PLoS One, Vol. 7, no. 1, e29839.
Rabb, G.B. (1994). The Changing Roles of Zoological Parks in Conserving Biological Diversity. American Zoologist, Vol. 34, no. 1, pp. 159-164.
WWF. (2016). Living Planet Report 2016. Gland, Switzerland: WWF.
Kakapo conservation – grasping at straws or crowdfunding conservation icon?
Posted: May 2, 2017 Filed under: 2017, ERES525, Research Essay | Tags: conservation, Crowdfunding, Flagship species, Kakapo, new zealand., Triage Leave a commentBy Hannah Graham-Cox
The ever-increasing human population is pushing more and more species towards the brink of extinction. With over 600 endangered species, New Zealand is struggling to prioritise ever decreasing funds from a stretched Department of Conservation (Kirk, 2015). So, how are these tough decisions reached? Many empirical methods have been used to assess whether a species is ‘worth’ conservation intervention. Some are simple and straightforward equations, while some are very convoluted involving many different variables. A novel term coming to the forefront as we realise that not all species can be saved, is triage. Triage in this sense, is the process of prioritising conservation activities; allocating scant resources to achieve maximum conservation returns (Bottrill et al., 2008).
Kakapo are an example of a species that may be designated a ‘lost cause’ if the triage approach were implemented by DOC. This nocturnal, flightless, extremely vulnerable bird was decimated by the combined efforts of human and invasive mammal predation, helped along by habitat loss. Now listed as ‘extinct in the wild’ by the IUCN red list, the only known kakapo are managed on pest free islands (Clout & Merton, 1998).
The history of kakapo is a sad and altogether too familiar one. Once, you could supposedly, “shake six from a single tutu bush” (Langton, 2000, p. 250). But following the arrival of humans and our mammalian co-invaders, the entire species was reduced to 51 individuals by 1995 (Ratley, 2014; Harrison & Moorhouse, n.d.). Helped along by modern technology and intensive management, the population has gradually climbed to just under 160 (News and updates from the Kākāpō Recovery Team, 2016). Kakapo have a limited role in ecosystem functioning mainly through vegetation, root and rhizome removal as well as contributing to seed dispersal through frugivory – mostly of rimu fruit (Clout & Hay, 1989; Gibbs, 2007; Atkinson & Merton, 2006). I have evaluated the general considerations when discussing the value of conserving a critically endangered species using kakapo as an example.
Role as flagship species
Flagship species are enigmatic or charismatic species which act as an ambassador for conservation in a given area or globally. Additional funds raised in the protection of this species may be allocated to other causes or conservation action, thus flagship species are often crucial for conservation (Simberloff, 1998; Bennett, Maloney, & Possingham, 2015). kakapo are what may be termed ‘high risk attention grabbers’ – they are a high profile species at great risk of extinction without immediate intervention. In order for species such as this to maintain public sympathy, results of conservation need to be rapid and tangible; for example, the positive response of kakapo to supplementary feeding in 1990-1991 which resulted in a significant population increase (Towns & Williams, 1993). Thus, it has been relatively straightforward to encourage funding from the general public and large corporations for Kakapo. The run-off effects of kakapo recovery include pest eradication and habitat restoration which benefits other species (Axed fund raises questions, 2012). Fortunately for kakapo, they are also charming and cheeky. A prime example of this is the ambassador for kakapo, Sirocco. Sirocco travels the country raising awareness and funds for the conservation of his species. Due to intensive hand rearing (he was the first male to be hand raised and they had not yet perfected avoiding human imprinting), it was realised that Sirocco had imprinted on humans and would not be a successful breeding bird but could make an excellent ‘public face’ for kakapo conservation (Sirocco, n.d.). After a botched attempt at mating with zoologist Mark Carwardine’s head was filmed and put on youtube, Sirocco shot to international fame. In 2011, two years after the infamous incident, 2000 people pre-ordered tickets to see Sirocco in Zealandia, raising funds for the sanctuary (Sirocco, n.d.).
Simply searching for ‘kakapo crowdfunding’ in Google brings up several campaigns raising funds for kakapo conservation. One example of this successfully raised the full goal of $45,400 which will go towards the kakapo 125 project which is described in the ‘Genetic Considerations’ section (Iorns, et al., 2016).
Endemism and phylogenetic uniqueness
An endemic species is found nowhere else on earth, kakapo are an example of such a species. They are described as ‘phylogenetically unique’ as they have been evolving in isolation from other related species for millions of years. Much value is placed upon those species which are rare or unique, and an increasing number of management programs prioritise these species. An example of this is the EDGE programme, launched by the Zoological Society of London, which stands for ‘Evolutionarily Distinct and Globally Endangered’ and prioritises conservation efforts on those species which are phylogenetically distinct as well as rare. As the only member of the subfamily Strigopinae, and being one of the world’s most endangered birds, kakapo rate very highly in the EDGE ranking system (EDGE : Bird Species Information – Kakapo, (n.d.).
Intrinsic value
The concept of the inherent value of nature can be traced back to Soulé who states that: ‘biotic diversity has intrinsic value irrespective of its instrumental or utilitarian value’ (Soulé, 1985). Thus in themselves, kakapo, evolving in seclusion for millennia, have value. Humans are also sentimental creatures whose dualism of being a part of nature, yet remaining distinct from it has resulted in feelings of guardianship towards nature (Bromley, 2013). The Māori term for this is kaitiakitanga which, through the partnership with Ngai Tahu and honoring Treaty of Waitangi responsibilities, is a core component of the The Conservation Act 1987 (Kawharu, 2000; Treaty of Waitangi Responsibilities, n.d.). Described as a unique, amusing, and beautiful, kakapo are not a difficult species to love either. In the words of Douglas Adams: “The kakapo is a bird out of time. If you look one in its large, round, greeny-brown face, it has a look of serenely innocent incomprehension that makes you want to hug it and tell it that everything will be alright” (Adams & Carwardine, 2009).
Labour intensive
All known kakapo are heavily monitored on three offshore islands: Whenua Hou (Codfish Island), Anchor Island and Hauturu o Toi (Little Barrier Island). Listed in Table 1. are tasks the Kakapo Recovery team, made up of ten full-time Department of Conservation staff, perform in order to help boost the kakapo population.
|
Activity |
What this involves |
|
Supplementary feeding |
Food supplied to the ‘wild’ kakapo must be expertly adjusted: too little food and the kakapo may not breed at all or the female may lay few eggs, too much food and the females may produce too many male offspring. |
|
Ongoing predator control |
All pests (mostly kiore or Pacific rat) exterminated from the three islands which kakapo have been removed to. Anchor island is still prone to stoat invasions as it is within swimming distance of the mainland, so maintenance must be ongoing on this island. |
|
Artificial insemination |
It was found that females who have mated more than once in a breeding cycle produce more fertile eggs than those that only mate once. The team remove sperm from selected males (often those with rare genes or who haven’t had many offspring) and using it to simulate a mating event with a female who has already been mated with in that breeding cycle. |
|
Incubation and hand rearing of chicks |
41% of the current kakapo population have been hand raised. This is an intensive process as the egg requires constant, specialised care. Chicks are returned to the wild at 4 months and have a survival rate of 91%. |
|
Monitoring |
Each kakapo has a smart transmitter attached to it, sending out data about the health of the bird as well as nesting and mating information. If a nest is established, the kakapo team have rostered ‘night-shifts’ where they guard the eggs or chicks for several months while the female is feeding. |
|
Health checks |
Full check-up of the bird is performed including: weighing, taking blood samples, checking the transmitter and removing parasites. This occurs yearly for adult birds, daily for nestlings and every 2-6 weeks for chicks under two years old. |
Table 1. Shows the methods through which the Kakapo Recovery Team is attempting to grow the current kakapo population. Adapted from “In the wild” (n.d.).
Cost
Kakapo conservation carries a hefty price tag. While finding exact figures of funds raised and tracing their use in kakapo conservation is difficult, in 1990 DOC entered into a partnership with Rio Tinto, New Zealand Aluminium Smelters Ltd and Forest & Bird. This partnership had raised $3.5 million as of 2010 before Rio Tinto dropped out in 2012 (Guyton & Deal, 2010; Axed fund raises questions, 2012). Due to the very ‘hands on’ approach of their recovery detailed above, every cent of that $3.5 million is vital. Much of the rest of the Kakapo Recovery Team’s funding comes from companies such as Meridian Energy, who have recently agreed to a three year partnership assisting the kakapo recovery fund (A Plan for the Future, n.d.). But three years in the grand scheme of kakapo conservation is not long at all – the long term goal of kakapo conservation is “To restore the mauri (life-force) of kākāpō by having at least 150 adult females” (A Plan for the Future, n.d.). With the current population of less than 160 individuals in total, and incredibly slow and irregular breeding cycles, this could take several decades. As described in the ‘Role as Flagship Species’ section above, crowdfunding is a more recent method for the Kakapo Recovery Group to raise funds for conservation. As is their ‘Adopt a Kakapo’ concept where members of the public can pay $100-500 to ‘adopt’ one of 13 birds currently available on their website (http://kakaporecovery.org.nz/adopt-a-kakapo/).
Genetic Diversity
A species can be described as ‘genetically depauperate’ if it has low genetic diversity compromising its longevity as a species. Genetic diversity in the kakapo population is extremely low after experiencing such a severe bottleneck when the population was reduced to only 51 individuals. All but one of the population’s founders (Richard Henry: the only male from the Fiordland population to produce offspring) came from the same population. Described as a ‘ticking time bomb’, inbreeding depression is the increased likelihood of extinction and results when there is a sustained level of inbreeding due to low population numbers for several generations (Jamieson, Wallis, & Briskie, 2006). Further loss of genetic diversity is being actively managed by DOC through preventing closely related individuals from mating and via a programme termed the kakapo 125 project which aims to sequence the genome of all known living kakapo (Projects – Genome Sequencing – Kakapo, n.d.). Kuia (pictured below), the only daughter of Richard Henry, will be crucial for maintaining diversity in the population. This is because all other kakapo are from a Stuart Island population which had already been isolated prior to human settlement of New Zealand (White et al., 2015a). Despite the efforts already taken, the effects of inbreeding depression are already visible in the current kakapo population and are listed in Table 2.
Kuia, Richard Henry’s daughter, nesting (Thompson, 2016).
|
Symptom of inbreeding depression |
Effect on a population |
Evidence of this occurring in the kakapo population |
| Low fertility | High number of infertile eggs or high risk of miscarriage (in mammal populations) |
40% of all eggs laid since 1985 have been infertile (average parrot infertility is around 10-15%) (Clout & Merton, 1998; Artificial insemination, n.d.). |
|
Low hatch or chick survival rates |
High chick mortality, low recruitment (chicks surviving to adulthood and supplementing the population) |
20% of embryos dying early in development (Artificial insemination, n.d.) |
| Reduced resistance to disease, predation, environmental stress | Increased risk of extinction through predation, disease, parasite infection, environmental stress or events (earthquakes, floods etc.) |
There are currently fears that exudative cloacitis, a bacterial disease which results in infected birds being unable to breed, may become a serious issue in kakapo populations due to reduced fitness (evolutionary longevity) through low genetic diversity (White, et al., 2015b). |
Table 2. Showing the symptoms of inbreeding depression, their effects on populations (or species) and the evidence of this observed in the kakapo population.
Conclusion
It is well documented that humans value rarity, but has conservation reached the stage where we no longer have the luxury of conserving species just because we find them charming and charismatic? Or are species such as kakapo still contributing to conservation funding enough as flagship species that it outweighs their lack of ecosystem functioning? Although often seen as pernicious and fatalistic, the triage approach to conservation has its groundings in prioritisation of increasingly limited resources (Towns & Williams, 1993). With budget cuts to the Department of Conservation occurring almost yearly, we need to have systems in place which will mean that these species not only have funding now, but ongoing – and a department to ensure habitat enrichment & pest control persists. I am of the opinion that species such as the kakapo deserve to be protected purely for their own sake. They represent millennia of evolution and their naivety and fragility despite this are endearing factors alone. Perhaps none could sum this up as well as the legendary Don Merton (pictured below) who has been instrumental in their rescue: “They are our national monuments. They are our Tower of London, our Arc de Triomphe, our pyramids. We don’t have this ancient architecture that we can be proud of and swoon over in wonder but what we do have is something that is far, far older. No-one else has kiwi, no-one else has kakapo. They have been around for millions of years, if not thousands of millions of years. And once they are gone, they are gone forever. And it’s up to us to make sure they never die out.” Sentimental reasons aside, kakapo populations have increased by more than 200% since the 51 recorded in 1995 (Plumb, 2016). If this trajectory is maintained, funds will continue to be raised and the future of kakapo will be more secure.
Chris Smuts-Kennedy, John Cheyne and Don Merton with Mandy the dog. The kakapo is Jill, the second male captured in the Esperance Valley, Fiordland, 1974 (Department of Conservation, 1974).
References
A Plan for the Future. (n.d.). Retrieved March 27, 2017, from http://kakaporecovery.org.nz/a-plan-for-the-future/
Adams, D., & Carwardine, M. (2009). Last chance to see. London: Arrow Books.
Artificial insemination. (n.d.). Retrieved April 21, 2017, from http://kakaporecovery.org.nz/artificial-insemination/
Atkinson, I. A. E., Merton, D.V. (2006). Habitat and diet of kakapo (Strigops habroptilis) in the Esperance Valley, Fiordland, New Zealand. Notornis. 53;37–54.
Axed fund raises questions. (2012, September). Retrieved March 25, 2017, from http://www.stuff.co.nz/dominion-post/news/7716027/The-commercialisation-of-conservation
Bennett, J. R., Maloney, R., & Possingham, H. P. (2015). Biodiversity gains from efficient use of private sponsorship for flagship species conservation. Proceedings of the Royal Society of London B: Biological Sciences, 282(1805), 20142693.
Bottrill, M. C., Joseph, L. N., Carwardine, J., Bode, M., Cook, C., Game, E. T., … & Pressey, R. L. (2008). Is conservation triage just smart decision making?. Trends in Ecology & Evolution, 23(12), 649-654.
Bromley, A. (2013). A Part of Nature or Apart from Nature? New Professors Explore Human Responses to the Environment. Retrieved April 27, 2017, from https://news.virginia.edu/content/part-nature-or-apart-nature-new-professors-explore-human-responses-environment
Clout, M. N., & Hay, J. R. (1989). The importance of birds as browsers, pollinators and seed dispersers in New Zealand forests. New Zealand journal of ecology, 27-33.
Clout, M. N., & Merton, D. V. (1998). Saving the Kakapo: the conservation of the world’s most peculiar parrot. Bird Conservation International, 8(03), 281-296.
EDGE : Bird Species Information – Kakapo. (n.d.). Retrieved April 24, 2017, from http://www.edgeofexistence.org/birds/species_info.php?id=1946
Gibbs, G. (2007). Ghosts of Gondwana; The history of life in New Zealand. Nelson: Craig Potton Publishing.
Guyton, S., & Deal, J. (2010). Christmas comes early for kakapo. Retrieved April 27, 2017, from http://www.tbfree.org.nz/christmas-comes-early-for-kakapo.aspx
Harrison, M., & Moorhouse, R. (n.d.). Kakapo (Strigops habroptila). Retrieved March 26, 2017, from http://www.edgeofexistence.org/birds/species_info.php?id=1946
In the wild. (n.d.). Retrieved April 23, 2017, from http://kakaporecovery.org.nz/in-the-wild/
Iorns, D., Digby, A., Robertson, B., & Howard, J. (2016). Sequencing the genomes of all known kākāpō. Retrieved April 24, 2017, from https://experiment.com/projects/sequencing-the-genomes-of-all-known-kakapo?s=discover
Jamieson, I. G., Wallis, G. P., & Briskie, J. V. (2006). Inbreeding and endangered species management: is New Zealand out of step with the rest of the world?. Conservation Biology, 20(1), 38-47.
Kawharu, M. (2000). Kaitiakitanga: a Maori anthropological perspective of the Maori socio-environmental ethic of resource management. The Journal of the Polynesian Society, 109(4), 349-370.
Kirk, S. (2015). No recovery plan to bring 600 native species back from brink of extinction. Retrieved March 26, 2017, from http://www.stuff.co.nz/national/politics/69920422/no-recovery-plan-to-bring-600-native-species-back-from-brink-of-extinction
Langton, G. (2000). Mr Explorer Douglas John Pascoe’s New Zealand Classic [Revised by Langton, G.]. Christchurch, New Zealand: Canterbury University Press, p. 250.
News and updates from the Kākāpō Recovery Team. (2016, November). Retrieved March 25, 2017, from http://createsend.com/t/i-35D4E98116C3A980
Plumb, S. (2016). Critically endangered kakapo on the increase – National – NZ Herald News. Retrieved April 27, 2017, from http://www.nzherald.co.nz/nz/news/article.cfm?c_id=1&objectid=11754390
Projects – Genome Sequencing – Kakapo. (n.d.). Retrieved March 24, 2017, from https://www.geneticrescue.science/projects/genome-sequencing/kakapo
Ratley, N. (2014, July). Back from the brink of extinction. Retrieved March 25, 2017, from http://www.stuff.co.nz/southland-times/news/features/10313116/Boom-A-kakapo-in-the-night
Sirocco. (n.d.). Retrieved April 24, 2017, from http://kakaporecovery.org.nz/sirocco/
Simberloff, D. (1998). Flagships, umbrellas, and keystones: is single-species management passé in the landscape era?. Biological conservation, 83(3), 247-257.
Soulé, M.E. (1985). What is conservation biology? Bioscience, 35, pp. 727–734
Towns, D. R., & Williams, M. (1993). Single species conservation in New Zealand: towards a redefined conceptual approach. Journal of the Royal Society of New Zealand, 23(2), 61-78.
Treaty of Waitangi Responsibilities. (n.d.). Retrieved April 27, 2017, from http://www.doc.govt.nz/about-us/our-policies-and-plans/conservation-general-policy/2-treaty-of-waitangi-responsibilities/
White, D. J., Hall, R. J., Jakob-Hoff, R., Wang, J., Jackson, B., & Tompkins, D. M. (2015a). Exudative cloacitis in the kakapo (Strigops habroptilus) potentially linked to Escherichia coli infection. New Zealand veterinary journal, 63(3), 167-170.
White, K. L., Eason, D. K., Jamieson, I. G., & Robertson, B. C. (2015b). Evidence of inbreeding depression in the critically endangered parrot, the kakapo. Animal Conservation, 18(4), 341-347.
Images
Title image: De Roy, T. (n.d.). Kakapo [Photograph]. Retrieved April 27, 2017, from https://www.islandconservation.org/kakapo-population-gets-a-much-needed-boost/
Department of Conservation (1974). Chris Smuts-Kennedy, John Cheyne and Don Merton with Mandy the dog and Jill the kakapo [Photograph]. Retrieved April 24, 2017, from http://www.doc.govt.nz/news/newsletters/behind-the-scenes/archived-newsletters/spring-2014/
Thompson, T. (2016). Kuia, Richard Henry’s daughter, nesting [Photograph]. Retrieved April 27, 2017, from https://www.islandconservation.org/kakapo-population-gets-a-much-needed-boost/
Introducing Exotic Dung Beetles to New Zealand
Posted: May 2, 2017 Filed under: 2017, ERES525, Research Essay Leave a commentBy Laura Malin-Curry
I remember driving through the country 10 years ago complaining to my grandfather how boring farms were to look at. My grandfather then replied that “that boring farm consists a dynamic relationship, where the sun provides the grass energy to grow, which then feeds the cows that in turn feed us”. Farms are novel ecosystems, built to sustain human development and growth. Globally, agriculture covers around 40% of terrestrial land area1. Locally, agriculture accounts for two-thirds of New Zealand’s exports, valued over $14.8 billion2.
As New Zealand’s economy is largely built around agriculture, maintaining and improving agricultural productivity is a primary goal for most farmers3. Currently, New Zealand native, forest dwelling, dung beetles are maladaptive to deal with pastoral dung. Resulting in dung limiting the amount of pastoral land available for grazing, thus adversely affecting the productivity of a farm3. To mitigate pastoral dung the Dung Beetle Release strategy Group aim to release 11 additional species of dung beetles into New Zealand to enhance the sustainable production of farms3.
The introduction of dung beetles into New Zealand pasture ecosystems has been in consideration for over 40 years8. However, movement took off in 2008 after the establishment of the Dung Beetle Release Strategy Group by a group of farmers and interested investors3. For the Dung Beetle Release Strategy Group to receive permission and funding to release exotic dung beetles, they had to provide information that 1- the dung beetles will not pose a threat to New Zealand native ecosystems and 2- that introducing dung beetles will be economically viable9.
Approximately $600,000 was invested into ensuring the safety of New Zealand ecosystem health upon releasing dung beetles3. And in 2011 the Environmental Protection Authority approved the release for 11 species9. In February 2014 the Dung Beetle Release Strategy Group completed a total of 74 dung beetle releases over 7 regions3. It is still too early to understand the impact that introduced dung beetles have on New Zealand’s agriculture.
Numerous countries are already benefiting from introducing dung beetles onto their pastoral land, such as Australia, America and Brazil3. These successful introductions give New Zealand a good indication towards the benefits that dung beetles will have on New Zealand. These benefits include; increasing farms productivity3, less greenhouse gas emissions7, reduction of livestock being infested with parasitic worms6 and the clearing up of New Zealand waterways9.
Releasing dung beetles into New Zealand to compliment, and enhance a novel ecosystem is not directly considered restoration. However, non-direct effects of dung beetles, like clearing up our waterways9, can be. The release will also positively affect our understandings in introducing species outside of their natural range. Adding to the tool box of conservation, resulting in better informed decision makings around the purposeful movement of a species outside of their native range.
Even though farms are artificial ecosystem composed of exotic species, I believe it is in New Zealand’s best environmental9 and economic interest2 to release exotic dung beetles to mitigate the effect of pastoral dung. Dung beetles are the missing link in our agricultural ecosystem, complimenting exotic stock life. Benefits of dung beetle release have already been seen in other countries3. Dung beetle release is well researched and will positively affect our agriculture. Soon enough we will see these exotic dung beetles as an ingrained vital component to our agricultural ecosystems.
References
- FAO stat, 2010, FAO stat, http://faostat.fao.org/site/339/default.aspx(2010) (Accessed March 2017)
- Sarah Brazil, ed. (2008).New Zealand Official Yearbook. Statistics New Zealand. p. 357. ISBN 978-1-86953-717-3. The export figure includes agriculture, horticulture and forestry.
- Dung Beetle Release Strategory Group. (2017, March 20). Dung Beetles in New Zealand. Retrieved from Dung Beetle: http://dungbeetle.org.nz/orders/
- Department of Conservation. (2017, March 25). Predator Free New Zealand 2050 to be a massive team effort. Retrieved from the Department of Conservation: http://www.doc.govt.nz/news/media-releases/2016/predator-free-nz-2050-to-be-a-massive-team-effort/
- King, C. (1984).Immigrant killers: Introduced predators and the conservation of birds in New Zealand / Carolyn King. Auckland, N.Z.: Oxford University Press.
- Nichols, Spector, Louzada, Larsen, Amezquita, & Favila. (2008). Ecological functions and ecosystem services provided by Scarabaeinae dung beetles.Biological Conservation, 141(6), 1461-1474.
- Slade, Eleanor M., Riutta, Terhi, Roslin, Tomas, & Tuomisto, Hanna L. (2016). The Role of Dung Beetles in Reducing Greenhouse Gas Emissions From Cattle Farming.Scientific Reports, 6, Scientific Reports, 2016, Vol.6.
- Hughes, R. (1975). Introduced Dung Beetles and Australian Pasture Ecosystems. Papers Presented At A Symposium During The Meeting Of The Australia and New Zealand Association For The Advancement Of Science At Canberra In January 1975.The Journal of Applied Ecology, 12(3), 819-837.
- Environmental Protection Authority. (2017, May 1). The Dung Beetle Decision. Retrieved from Environmental Protection Authority: http://www.epa.govt.nz/new-organisms/popular-no-topics/dungbeetles/Pages/decision.aspx
Will climate change make our current system of nature reserves redundant?
Posted: May 6, 2016 Filed under: 2016, ERES525, Research Essay, Uncategorized | Tags: assisted migration, climate change, range shift, reservation Leave a commentBy Amanda Healy
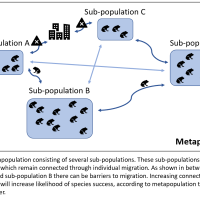
Ecological reservation is currently used as a primary technique for preserving species or ecosystems. By disallowing the exploitation of an ecosystem, it is assumed that the area will be protected, and will therefore be able to exist into perpetuity. However, due to the rapidly increasing temperatures caused by anthropogenic climate change, many different species are moving away from their previous ranges into more climatically suitable locations (Chen et al., 2011; Loarie et al., 2009). This essay will look at how that may affect ecological reserves, and what we may need to do to keep up with the ever-changing climate.

Images showing predictions for global climate change in the coming years. From express.co.uk
Climate-change induced range shifts are occurring in a vast number of species (Shoo et al.,2006). One study found that on average, species are moving to higher latitudes and altitudes at rates of 16km and 11m per decade, respectively (Chen et al., 2011). These rates obviously vary, depending on the intensity of climate change in any given area and the ranging ability of the species in question; migratory species are able to shift their ranges quickly, but sedentary species (such as trees) take much longer (Parmesan et al., 1999).
Because of the movement of species out of their original ranges, our current system of protected reserves may become redundant in the future. One estimate states that in 100 years, only 8% of our reserves will still have the same climate as they have today (Loarie et al., 2009). This means that many of the species that we are aiming to protect will no longer be able to live within these reserves. They will either move outside of the reserve’s borders, or even worse, barriers will inhibit their movement and they will go locally extinct.
The protection of these reserved species will likely require assisted colonisation in the future (Lunt et al., 2013). The barriers that inhibit the movement of species, such as habitat fragmentation or the fencing around reservations, mean that these species will need help to move to a habitat that is suitable in the changing climate. The same applies to species that are slow moving or sedentary, as they are unlikely to be able to keep pace with the rate of climate change (Parmesan et al., 1999). This concept goes against traditional ideas of conservation and reservation, as it would often mean introducing a species to a geographical area that they have never occupied previously (Hoegh-Gulberg et al., 2008). Most reservations work to preserve only species that are native to the area. However, in order to save many of these species, it will likely be the best option in the coming years.
For these reasons, it is likely that nature reserves, for the purpose of species or ecosystem preservation, have a limited lifespan. At some point, as temperatures continue to rise and climates continue to move, we will have to reconsider our concepts of reservation ecology. Alternative solutions will need to be considered in order to protect the organisms that these reserves are currently housing.
References
Chen, I. C., Hill, J. K., Ohlemüller, R., Roy, D. B., & Thomas, C. D. (2011). Rapid range shifts of species associated with high levels of climate warming. Science, 333(6045), 1024-1026.
Hoegh-Guldberg, O., Hughes, L., McIntyre, S., Lindenmayer, D. B., Parmesan, C., Possingham, H. P., & Thomas, C. D. (2008). Assisted colonization and rapid climate change. Science (Washington), 321(5887), 345-346.
Loarie, S. R., Duffy, P. B., Hamilton, H., Asner, G. P., Field, C. B., & Ackerly, D. D. (2009). The velocity of climate change. Nature, 462(7276), 1052-1055.
Lunt, I. D., Byrne, M., Hellmann, J. J., Mitchell, N. J., Garnett, S. T., Hayward, M. W., … & Zander, K. K. (2013). Using assisted colonisation to conserve biodiversity and restore ecosystem function under climate change.Biological conservation, 157, 172-177.
Parmesan, C., Ryrholm, N., Stefanescu, C., Hill, J. K., Thomas, C. D., Descimon, H., … & Tennent, W. J. (1999). Poleward shifts in geographical ranges of butterfly species associated with regional warming. Nature,399(6736), 579-583.
Shoo, L. P., Williams, S. E., & Hero, J. (2006). Detecting climate change induced range shifts: Where and how should we be looking? Austral Ecology, 31(1), 22-29.
Willis, S. G., Hill, J. K., Thomas, C. D., Roy, D. B., Fox, R., Blakeley, D. S., & Huntley, B. (2009). Assisted colonization in a changing climate: a test‐study using two UK butterflies. Conservation Letters, 2(1), 46-52.
Should the Ability for Restoration Justify the Degradation, Damage, or Destruction of Environments?
Posted: May 4, 2016 Filed under: 2016, ERES525, Research Essay | Tags: #restoration, Destruction, invasive species, mining, native species, Powelliphanta Leave a commentBy Olivia Quigan
Restoration is becoming an increasingly useful tool in conservation. We can now bring biodiversity back to an area that has been impaired beyond recognition by human activities, such as logging, damming, or open cast mining. Given that we are restoring more and more ecosystems around the world, does this give us leave to destroy ‘pristine’ habitats in order to exploit them to gain access to resources?
The benefits of destroying habitats in order to access resources are mostly of economic value. When cost-benefit analyses for an open cast mine are done, the only environmental outcomes that are considered are those that can be turned into a monetary value (Abelson, 2015). These are quantified as the physical impacts on the environment and how these impact health and agriculture (Abelson, 2015). This ignores the intrinsic value of unique species, as through losing them we reduce global biodiversity – a value that cannot be measured in currency (Campbell, 2014).
An argument could be made for destruction of habitats with a view to restoration, that given enough planning time, species can be saved before the habitat loss occurs. They could then be returned to the habitat during the restoration process, or found suitable homes elsewhere, that are similar to their current habitat. Translocation can be a valid restoration method, however not without its risks. This method was attempted in 2011. To allow for an open cast mine in New Zealand’s South Island, the unique and endemic Powelliphanta augusta Snails were collected and stored in shipping containers with the ultimate goal of introducing them to nearby forests. This resulted in the deaths of 800 individuals due to a technical failure of the refrigeration unit they were stored in (Vallance, 2011). This attempt was a failure, because even with the remaining snails being translocated, they are not
successfully persisting in their new environment, with death rates at new sites of up to thirty per cent (Morris, 2010). There are many other instances of failed translocations. Analyses of many reptile and amphibian translocations between 1991 and 2006 showed that up to 30 per cent of translocations failed in producing self-sustaining populations (Germano & Bishop, 2008). This rate of failure must force us to come to the conclusion that we do not yet have the knowledge to prevent extinctions in the case of a planned environmental degradation, especially when endemic species are involved, as the risk is often too high to justify needless environmental degradation.
The creation of novel landscapes is an inevitable outcome of the anthropogenic influence on the world. As we remove natural habitats, the areas that replace them won’t be the same; no matter how hard we try to restore them. A study by Lugo, Carlo and Wunderle Jr. (2011) looked at the islands of Puerto Rico and the introduced species there.
The forest cover here dropped from 100 per cent to just six per cent by the 1940s. The restoration of much of the forest has included many introduced species, both plants and animals. The resulting forest was a mixture of both, but the native plant species continue to dominate the forests, with cover of over 80 per cent. Native birds continue to be successful, and forage on both natives and introduced plants. The introduced honeybee appears to have adapted to the phenology of the native plants and is an important pollinator (Lugo, Carlo, & Wunderle Jr., 2012). This indicates that novel habitats created by restoration efforts can be sustained with introduced species, but we must continue to protect the native species to ensure lasting intrinsic value of the ecosystem.
Disturbed habitats are more likely to be susceptible to invading species. These are defined as species which “proliferate and noticeably replace native species,” (Clewell & Aronson, 2013). Invasive species with a more generalist way of life will have an advantage over native species, especially if these species have evolved into a more specialist niche (Clewell & Aronson, 2013). This is even more applicable in island habitats, where animals have evolved with limited predators. Using the land for agriculture or industry changes the scope of the ecosystem, and increases the vulnerability of it to invasions from non-native species (Vitousek, D’Antonio, Loope, Rejmanek, & Westbrooks, 1997). Human modification of environments is a major driver the invasion by non-native species. Logged forests in Thailand that were home to an invasive weed experienced an eight-fold reduction in pollinators visiting native species. The invasive beetle, Coccinella septempunctata, showed higher abundances in agricultural grasslands when compared to non-modified areas (Didham, Tylianakis, Gemmell, Rand, & Ewers, 2007). Due to the precious value of native species, the total destruction of a habitat cannot be justified as this disturbance leads to increased vulnerability to species invasions.
The complete destruction of a habitat will always be detrimental to the species living there. To destroy a habitat for monetary gain is to place a value on the uniqueness of habitats, and deem it less important than the economy. The evidence shows that we are not capable of maintaining the integrity of a habitat if we destroy it completely. Disturbed habitats are more likely to allow invasive species, which decimate native populations. Human attempts at preservation by translocation of species often fail. As we cannot guarantee the safety of our unique species, we cannot justify the destruction of any habitat; regardless of how accomplished we are becoming at restoring them.
References:
Abelson, P. (2015). Cost–Benefit Evaluation of Mining Projects. The Australian Economic Review, 442-52.
Campbell, R. (2014). Seeing through the dust: Coal in the Hunter Valley Economy. Canberra: The Australia Institute.
Clewell, A. F., & Aronson, J. (2013). Ecological Restoration – Principles, Values & Structure of an Emerging Profession (2nd ed.). Washington, D.C: Island Press.
Didham, R. K., Tylianakis, J. M., Gemmell, N. J., Rand, T. A., & Ewers, R. M. (2007). Interactive effects of habitat modification and species invasion on native species decline. Trends in Ecology and Evolution, 22(9), 489-496.
Germano, J. M., & Bishop, P. J. (2008). Suitability of Amphibians and Reptiles for Translocation. Conservation Biology, 7-15.
Lugo, A. E., Carlo, T. A., & Wunderle Jr., J. M. (2012). Natural mixing of species: Novel plant-animal communities on Caribbean Islands. Animal Conservation, 233-241.
Morris, R. (2010). An Unfortunate Experiment. Forest And Bird, 14-16.
Vallance, N. (2011, November 10). Snail fridge deaths an avoidable tragedy. Retrieved from Forest and Bird: http://www.forestandbird.org.nz/what-we-do/publications/media-release/snail-fridge-deaths-avoidable-tragedy
Vitousek, P. M., D’Antonio, C. M., Loope, L. L., Rejmanek, M., & Westbrooks, R. (1997). Introduced Species: A significant component of Human-caused Global Change. New Zealand Journal of ecology, 1-16.
Cultural Ecosystem Services and Restoration: Reconnecting communities and people with nature
Posted: May 4, 2016 Filed under: 2016, ERES525, Research Essay | Tags: community connections, cultural ecosystem services, ecological restoration, people and nature Leave a commentCultural Ecosystem Services and Restoration: Reconnecting communities and people with nature
By Andrea Hirschberg
As people realise how degraded the environment has become, more are turning to local ecological restoration projects to help ‘do their bit’. Greater Wellington alone has over 30 local community based restoration groups listed on its web page (GWRC, 2016), with likely many more unlisted. For many restoration groups the aim is to restore the physical environment or return a particular species to the area. However, for other groups the cultural aspect (such as community connections and education) of restoration is the main aim of the project (Fernandez-Gimenez et al., 2008).
Cultural Ecosystem Services
Saeukhan and Whyte (2005) describe cultural ecosystem services (CES) as “nonmaterial benefits people obtain form ecosystems through spiritual enrichment, cognitive development, reflection, recreation and aesthetic experiences”. While CES are often highly regarded by many who are participating in restoration projects (Brancalion et al., 2014). They are however, often not covered in a lot of ecosystem services research (Chan et al., 2012 b) and there is currently poor integration of CES into management plans (Milcu et al., 2013, Plieninger et al., 2012 & Chan et al., 2012 a). The aim of this essay is to look at how restoration and cultural ecosystem services can foster community connections and connections between the people and the land
As Milcu et al. (2013) found, with the exception of recreational, aesthetic, heritage and educational services, there is very little inclusion of cultural ES into management plans. Meaning that values such as spiritual value, cultural identity and history and the knowledge system (Tilliger et al. 2015) in relation to the ecosystem are often left out of management plans. This means that many management plans lack the full range of success indicators available to them. A broader range of social-science tools need to be used when putting together a management plan to include cultural values rather than just economic values (Chan, et al. 2012 b & Tilliger et al. 2015). Tilliger et al. (2015) believe that the lack of inclusion and study of CES is due to CES being less tangible than other ES and often including non-use values making CES harder to estimate and quantify.
Interconnections of People and Nature
Community-based natural resource management can play a significant role in ecological restoration projects. This is done by providing civic engagement through resource and knowledge pooling, the growth of trust between stakeholders and connection with other community groups (Hibbard et al. 2006). The strengthening of relationships between community members was found to be an important factor for members of restoration groups by Kittinger et al. (2013) and Fernandez-Gimenez et al. (2008). One member of Kittinger et al. (2013) study states “we are not just restoring an ecosystem but a community”. In their study looking at collaborative, community-based forestry organisations Fernandez-Gimenez et al. (2008) found that this community building aspect of the restoration project was the most important aspect for some members of the restoration group. The restoration project provided a space for those who were interested in the same place to learn together and share knowledge about that place. Community based restoration projects allow a diverse range of people to come together for a common purpose and create a plan which relevant to them.
Fernandez-Gimenez et al. (2008) also found that restoration projects helped to reconnect people with the land and engaged people in the natural resources around them. Participating in community restoration projects helps people become more aware of the interconnectedness of nature and how their action affects the environmental health (Egan et al. 2011 and Kittinger et al. 2013). This is especially true for restoration projects based on traditional ecological knowledge (TEK). Indigenous communities tend to have a more holistic world view than western science (WS) and many indigenous communities see themselves as a part of nature and on an equal level to everything else that makes up the ecosystem. For Maori this holistic world view has resulted in the idea of mauri (or life force of something). In terms of restoration, this means that if the mauri of the land is damaged then the mauri of the people is also damaged; if the land is sick the people are as well. Maori also believe in kaitiakitanga where everyone is a guardian of the land and everyone has responsibility to maintain the mauri of the land (Henwood & Henwood & Roberts et al. 1995). These two values are often what underpin iwi, hapu and whanau based restoration projects and are instrumental in reconnecting people with their land.
In their 2015 study Tilliger et al. focused on the connections between CES and the connections between CES and the land. They found that as cultural values and cultural connections to the land were lost degradation of the land occurred, which in turn resulted in a further loss of CES from the land, as shown in FIG. 1 below. I believe that in restoration projects the reverse can be true; as the land is restored CES will increase which will increase the restoration efforts.
 |
| Figure 1. Shows the connections between CES and the land and how a reduction in one can result in a reduction of the other. From Tilliger et al. 2015 |
Conclusion
While there is currently a lack of studies and restoration management plans which focus on CES (Chan et al. 2012 b & Tilliger et al. 2015) those studies which have looked at CES (including Brancalion et al. 2014, Kittinger et al. 2013 , Fernandez-Gimenez et al. 2008 and Milcu et al. 2013) found that CES are widely regarded by participants. In some cases the reconnection of communities was the main reason for many participants becoming involved (Kittinger et al. 2013 and Fernandez-Gimenez et al. 2008). The reconnection of mana whenua with the land and the reassertion of kaitiakitanga by the mana whenua is often the driving factor of Maori led restoration projects in New Zealand (Henwood & Henwood & Roberts et al. 1995)
References
Brancalion, P.H.S, I. Villarroel Cardozo, A. Camatta, J. Aronson & R.R. Rodrigues, 2014. Cultural Ecosystem Services and Popular Perceptions of the Benefits of an Ecological Restoration Project in the Brazilian Atlantic Forest. Restoration Ecology 22 (65-71)
Chan, K.M.A., Satterfield, T., & Goldstein, J., 2012(a). Rethinking ecosystem services to better address and navigate cultural values Ecological Economics 74 (8-18)
Chan, K.M.A, A.D. Guerry, P. Balvanera, S. Klain, T. Satterfield, X. Basurto, A. Bostrom, R. Chuenpagdee, R. Gould, B.S. Halpern, N. Hannahs, J. Levine, B. Norton, M. Ruckelshaus, R. Russel, J. Tam & U. Woodside, 2012 (b). Where are Cultural and Social in Ecosystem Services? A Framework for Constructive Engagement. BioScience 62 (744-756)
Egan, D., Hjerpe, E.E., & Abrams, J. (eds). 2011. Human dimensions of ecological restoration: Intergrating science, nature and culture. Island Press, Washington DC. 410pp.
Fernandez-Gimenez, M.E., Ballard, H.L. & Sturtevant, V.E., 2008. Adaptive Management and Social Learning in Collaborative and Community-Based Monitoring: a Study of Five Community-Based Forestry Organizations in the western USA. Ecology and Society 13 (2)
Greater Wellington Regional Council 2016. www.gw.govt.nz/local-care-groups/ Accessed on 29th March 2016
Hibbard, M. & Lurie, S. 2006. Some community socio-economic benefits of watershed councils: A case study from Oregon, Journal of Environmental Planning and Management, 49(6), 891-908
Kittinger, J.N., Bambico, T.M., Minton, D., Miller, A., Mejia, M., Kalei, N., Wong, B., & Glazier, E.W. 2016. Restoring ecosystems, restoring community: socioeconomic and cultural dimensions of a community-based coral reef restoration project, Reg Environmantal Change, 16, 301-313
Milcu, A. Ioana, J. Hanspach, D. Abson, and J. Fischer, 2013. Cultural ecosystem services: a literature review and prospects for future research . Ecology and Society 18(3)
Plieninger, T., Dijks, S., Oteros-Rozas, E., & Bieling, C. 2013. Assessing, mapping, and quantifying cultural ecosystem services at community level. Land use Policy 33, 118-129
Sarukhan, J., & Whyte, A., (eds). 2005. Ecosystems and human well-being: Synthesis (Millennium Ecosystem Assesment). Island Press, Washington DC.
Shandas, V. & Messer, W.B, 2008. Fostering Green Communities Through Civic Engagement: Community-Based Environmental Stewardship in the Portland Area, Journal of the American Planning Association, 74:4, 408-418
Tilliger, B., Rodriguez-Labajos, B., Bustamante, J.V., & Settele, J., 2015. Disentangling values in the interrelations between cultural ecosystem services and landscape conservation-A case study of the Ifugao Rice Terraces in the Philippines. Land 4, 887-931
The SeaWorld Controversy: Not so Black and White
Posted: May 4, 2016 Filed under: 2016, ERES525, Research Essay | Tags: aquariums, conservation, SeaWorld, World Association of Zoos and Aquariums, zoos Leave a commentLooking Into Zoos and Aquariums and the Controversies Behind Them
By Brenda Perez
When I was a little kid, I remember going to the aquarium and being mesmerized by all the different sea creatures. In the sixth grade, I told my friends that I wanted to be a marine biologist so that I would be able to work with sea animals. I distinctly remember my friend teasing me and telling me that I would be one of the people training and doing shows with the dolphins. At age 12, that sounded like a dream. However, ten years later I have achieved my dream of becoming a marine biologist, but have chosen not to pursue the path of being a dolphin trainer. As a child, or even as an adult coming from a non-scientific viewpoint, you don’t tend to think about all the negative aspects that come with not only theme parks with animals, but all zoos and aquariums. As I have gone through life and school, I have a greater understanding of marine biology and conservation which has lead me to consider both advantages and disadvantages of these situations. When thinking about controversies such as SeaWorld, one has to go further and look at the positive and negative aspects of aquariums and zoos in general.
When is it conservation and when is it cruelty: The good, the bad, and the compromise
Zoos and aquariums provide researchers with the ability to study the behavior of animals in their “natural” environment (Ballanthyne et al. 2007). They frequently house the last individuals of the most threatened species around the world (Clarke 2009) and act not as a replacement for saving animals, but as a last resort or “holding area” for endangered species due to the fact that their native habitats are uninhabitable (Conway 2011). Captive breeding is also used in an effort to stabilize the species to a point where they will be able to sustain themselves in the wild (Hutchins et al. 2003).
Additionally, zoos and aquariums help educate the public about several things that they would otherwise not normally be exposed to. By visiting these facilities, people can learn about how global warming affects animals and environments, biodiversity issues facing species (Kawata 2013), and specific issues facing animals in their region (Whitham and Wielebnowski 2013). People get to connect with animals on a

Image 1: Pamphlet given out at Zoo Atlanta with information about endangered species and conservation efforts. (Zoo Atlanta)
personal level and are exposed to not only environmental education, but also conservation strategies (Image 1) and what they can do to help (Patrick et al. 2007). They get emotionally engaged and become more open to communication about conservation both locally and worldwide (Ballantyne et al. 2007). After their visits to aquariums and zoos, people recognized that they could be a part of the solution to environmental problems by taking action in conservation efforts. Visitors believe that zoos and aquariums play an important role in animal care and conservation education and left feeling a stronger connection to nature (Falk et al. 2007; Heimlich et al. 2005). Many people in the central regions of countries would not be exposed to marine issues if it weren’t for aquariums, and they, along with zoos, provide additional insight on issues facing animals and environments globally. However, these are aspects that people do not initially take into account when thinking about zoos and aquariums.
SeaWorld
When you ask most children what they think of SeaWorld, their immediate response is excitement about all of the amazing animals they have there. However, when you ask many adults, their first instinct is skepticism and resentment towards the conditions of said animals.
SeaWorld is criticized heavily by the public for several reasons: forcing its animals to put on shows for audiences, keeping them in tanks that are far too small, capturing animals from the wild, and separating families. But the public has been largely swayed by the media, which tends to focus on the negatives, as well as the “documentary” Blackfish, a persuasive piece that looks at only one side of the situation and appeals to human emotions. Granted, Blackfish brings these up as valid points but they do so in a manipulated context (Pierce 2015). In SeaWorld, killer whale calves are kept with their mothers and whales haven’t been captured from the wild in over 35 years. All of the animals that reside in any of their parks are taken care of physically and well treated (Walsh et al. 1994). They are studied for animal research in ways that scientists are unable to achieve in the wild (Falcato 2016). SeaWorld has rescued over 27,000

Image 2: Children are exposed to and learn about killer whales and the importance of conservation efforts. (SeaWorld)
animals and many of them have been returned to the wild after rehabilitation (Parham 2001). Busch Gardens, a SeaWorld park, has several birds, reptiles, and mammals on display at each location. Busch Gardens Tampa Zoo alone has over 12,000 animals including 250 species, of which more than 30 are threatened or endangered. In a little over ten years, the SeaWorld and Busch Gardens Conservation Fund has given more than $10 million to over 700 projects around the world (Pierce 2015). Additionally, SeaWorld offers camps for children which educate them about all aspects of sea life and gets them involved from an early age (Image 2). While we may not agree with all aspects of SeaWorld, we have to realize that there are a great number of benefits that happen behind the scenes.
Aquariums and Zoos
Zoos and aquariums get criticized for the stress that they may cause to their animals (Morgan and Tromborg 2007). Touch tanks can be found at several aquariums nowadays to provide the possibility of personal interaction for visitors. The argument could be made that by allowing countless individuals to touch these animals that it could cause them stress. However, most animals that are exposed to this experience are more resilient; brittle stars, sea urchins, sand dollars, and horseshoe crabs have tough, rigid exteriors that can tolerate being handled. In addition, these animals are cycled throughout the day in order to limit the amount of time they are exposed (Rowe and Kisiel 2012). In addition to the stress of being handled, zoos and aquariums are criticized for having their animals exposed to artificial lighting, loud or sudden noises, uncomfortable temperatures, modified feeding schedules, and a limited living area. What people fail to realize is that caretakers do not simply haphazardly assign animals to spaces and forget about them. Animal behaviors are studied and recorded in order to provide better care and maintenance for each animal and its habitat. Feeding schedules are developed specifically for animals with their health as a first priority (Morgan and Tromborg 2007). Yes, different locations have different sizes of tanks or enclosures with varying amounts of animals in each of them. What many people tend to focus on is overcrowding within one area or limited space for the larger animals (Heimlich et al. 2005). Sadly, in the United States, about 90% of aquariums and zoos have not been accredited by the Association of Zoos and Aquariums (AZA), meaning that they do not comply with the standards set by the accreditation commissioners (Association of Zoos and Aquariums 2010). The AZA has specific guidelines for all animals and the conditions that they should be kept in. there are approximately 2,100 aquariums and zoos in the US that are not AZA accredited. In many cases, there are financial limitations preventing expansion programs. Occasionally, these conditions can lead to animal conflict, or even deaths (Hutchins 2006), and standards that many would not consider suitable. The New Zealand National Aquarium houses a solitary Hawksbill sea turtle in a tank only seven times as long and two times as wide as it. He has been there for 27 years having being born in captivity. Not having been accredited by the World Association of Zoos and Aquariums, the standards of living for this poor animal are not nearly where they should be (World Association of Zoos and Aquariums 2005). While one can argue that the educational and conservation benefits of all aquariums and zoos, we cannot ignore that in some instances non-accredited aquariums may have been detrimental.
At the same time, aquariums and zoos have great rehabilitation programs for animals, including seals, sea turtles, dolphins, frogs, birds, wolves, monkeys and many more, that work behind the scenes and out of the public eye. These programs have helped nurse countless animals back to health and have successfully returned them back to their homes in the wild (Image 3; Rakes et al. 1999). Breeding programs exist to replenish populations and keep endangered animals from becoming extinct (Hutchins et al. 2003). If it weren’t for the conservation programs affiliated with aquariums and zoos, many of these animals would continue to decrease in the wild. By having programs that inform the public about these issues, there is an increased awareness of these issues and that has lead people to be more proactive in these fields. (Gross 2015).
World Association of Zoos and Aquariums
One of the most prominent members of WAZA is America’s AZA. Established in 1924, AZA’s goals include conserving wild animals, reintroducing endangered species, and restoring habitats. The accreditation commission has strict guidelines for the species they house: each animal’s enclosure or tank must meet living conditions and dimensions that vary with size and amount of individuals. The association has Animal Care Manuals for each species that get updated regularly

Image 4: One of the many Animal Care Manuals followed by the AZA. (Association of Zoos and Aquariums)
and must be followed in order to maintain accreditation (Image 4). For example, a zoo or aquarium cannot keep animals in captivity if they aren’t considered a good candidate (Association of Zoos and Aquariums 2010). The AZA SAFE: Saving Animals From Extinction Program is focusing on the following ten endangered species with the goal of engaging the public to promote conservation: African penguins, Asian elephants, black rhinoceros, cheetahs, gorillas, sea turtles, sharks, vaquitas, Western pond turtles, and whooping cranes (Colbert 2016).
Currently, 233 zoos and aquariums have been accredited by AZA in the US. While a small victory, that is sadly only about 10% of all zoos and aquariums in the states. Nevertheless, these institutions hold 750,000 animals representing 6,000 species, of which 1,000 are endangered (Colbert 2016). These animals impact 180 million people annually. Each year, AZA provides $160 million to about 2,700 conservation projects in 115 countries over the world. Last year, they partnered with 575 nonprofit, government, and private organizations for these projects (Colbert 2016).
The AZA is one of 22 association members of the World Association of Zoos and Aquariums. Since 1946, WAZA has included several different associations all over the world that follow the same accreditation standards as those enforced by AZA. WAZA includes more than 330 zoos and aquariums over 50 countries. More than 700 million people visit their accredited facilities all over the world annually (World Association of Zoos and Aquariums 2005). Because of WAZA accredited zoos and aquariums, hundreds of thousands of animals are being properly cared for with appropriate living conditions; all over the world environmental education and conservation efforts are increasing. And that is amazing.
Conclusion
It would be misleading to characterize zoos and aquariums in either a fully positive or negative light. At an emotional level, many of us might want them to not exist and have all the animals be free to live their lives out in the wild. However, the sad truth is that many animals would not be able to survive in the wild without the chance they have had to grow as a population or the care that they are currently given in zoos and aquariums. While there can be no perfect harmony or solution, it appears that the best solution is to strive to have more facilities become accredited by WAZA.
References
Association of Zoos and Aquariums. (2010). The accreditation standards and related policies.
Ballantyne , R., Packer, J., Hughes, K., & Dierking, L. (2007) Conservation learning in wildlife tourism settings: lessons from research in zoos and aquariums, Environmental Education Research, 13(3), 367-383
Clarke, A. G. (2009). The Frozen Ark Project: the role of zoos and aquariums in preserving the genetic material of threatened animals. International zoo yearbook, 43(1), 222-230.
Colbert, D. “AZA SAFE: Engaging People through Education” (2016).
Conde, D. A., Flesness, N., Colchero, F., Jones, O. R., & Scheuerlein, A. (2011). An emerging role of zoos to conserve biodiversity. Science,331(6023), 1390-1391.
Conway, W. G. (2011), Buying time for wild animals with zoos. Zoo Biology, 30: 1–8.
Falcato, J. (2016). “Thematic Aquariums – The Right Approach?” Der Zoologische Garten 85(1-2): 14-25.
Falcato, J. (2016). “The modern zoo – How do people perceive zoo animals” Applied Animal Behaviour Science 85(1-2): 14-25.
Falk, J.H., Reinhard, E.M., Vernon, C.L., Bronnenkant, K., Deans, N.L.; Heimlich, J.E., (2007). Why Zoos & Aquariums Matter: Assessing the Impact of a Visit. Association of Zoos & Aquariums. Silver Spring, MD.
Gross, M. (2015). “Can zoos offer more than entertainment?” Current Biology 25(10): R391-R394.
Heimlich, J., Bronnenkant, K., Barlage, J., & Falk, J. H. (2005). Measuring the learning outcomes of adult visitors to zoos and aquariums: Phase I study. Bethesda, MD: American Association of Zoos and Aquariums.
Hutchins, M. (2006). Death at the zoo: the media, science, and reality. Zoo Biology, 25(2), 101-115.
Hutchins, M., Smith, B., & Allard, R. (2003). In defense of zoos and aquariums: the ethical basis for keeping wild animals in captivity. Journal of the American Veterinary Medical Association, 223(7), 958-966.
Kawata, K. (2013). “Rambling Thoughts on Zoo Animal Collection and Conservation: A Historical Perspective.” Der Zoologische Garten 82(1-2): 26-39.
Morgan, K. N. and C. T. Tromborg (2007). Sources of stress in captivity. Applied Animal Behaviour Science 102(3-4): 262-302.
Parham, D. (2001). To the Rescue!: The SeaWorld/Busch Gardens Animal Rescue and Rehabilitation Program. SeaWorld Education Department.
Patrick, P. G., Matthews, C. E., Ayers, D. F., & Tunnicliffe, S. D. (2007). Conservation and education: Prominent themes in zoo mission statements. The Journal of Environmental Education, 38(3), 53-60.
Pierce, A. (2015). SeaWorld and the Blackfish: The Lack of Two-Way Symmetrical Communication and Finding the Correct Message (Doctoral dissertation, California Polytechnic State University, San Luis Obispo).
Rakes, P. L., Shute, J. R., & Shute, P. W. (1999). Reproductive Behavior, Captive Breeding, and Restoration Ecology of Endangered Fishes. Environmental Biology of Fishes 55(1), 31-42.
Rowe, S., & Kisiel, J. (2012). Family engagement at aquarium touch tanks—Exploring interactions and the potential for learning. In Understanding interactions at science centers and museums (pp. 63-77).
Walsh, M. T., Cambell, T. W., Phillips, B., Kerivan, J. M., & Davis, R. L. (1994). Medical care of stranded sea turtles at SeaWorld of Florida. InProceedings of the Thirteenth Annual Symposium on Sea Turtle Biology and Conservation. US Department of Commerce, National Oceanic and Atmospheric Administration, National Marine Fisheries Service, Miami (Vol. 189).
Whitham, J. C. and N. Wielebnowski (2013). New directions for zoo animal welfare science. Applied Animal Behaviour Science 147(3-4): 247-260.
World Association of Zoos and Aquariums. United for Conservation. (2005).Building a future for wildlife: The world zoo and aquarium conservation strategy. WAZA Executive Office.
Restoring resilience: Can restoring coasts with ecosystem-based solutions protect social-ecological systems from the impacts of climate change?
Posted: May 2, 2016 Filed under: 2016, ERES525, Research Essay | Tags: #resilience, climate change, coastal hazard, ecosystem-based defence, restoration Leave a commentBy Anni Brumby
Victoria University of Wellington
Background
The destruction of hurricane Katrina in New Orleans in 2005 (Photo 1), extreme flooding on the east coast of Australia in 2007, and last year, my local train station in Porirua completely underwater. Welcome to the stormy and wet world of global climate change.
Many of the threats caused by climate change are especially severe in coastal and low lying areas (Nicholls et al., 2007). This is a major concern, as coasts all over the planet are densely populated. Coastal areas less than 10 metres above sea level cover only 2% of the Earth’s surface, but contain 13% of the world’s urban population (McGranahan, Balk, & Anderson, 2007). Often coasts are highly modified for human purposes, and crucial for economic stability (Martínez et al., 2007).
The observed and predicted coastal hazards include sea level rise and the resulting inundation; erosion and salinization of land (Gornitz, 1991); increased precipitation intensity and run-off; and storm flooding (Nicholls & Lowe, 2004). Climate change will also increase the frequency and intensity of weather extremes, such as hurricanes (Emanuel, 2005; Seabloom, Ruggiero, Hacker, Mull, & Zarnetske, 2013).
The existence of Homo sapiens rely on ecosystem services – “the benefits people obtain from ecosystems” (Millennium Ecosystem Assessment, 2005, p. 1), such as food production, raw materials, waste treatment, disturbance and climate regulation, water supply and regulation…The list goes on. Coastal ecosystems contribute 77% of global ecosystem-services value (Martínez et al., 2007), thus any coastal threats affect have major impacts for humans both economically and socially.
It is unlikely that we can stop global warming (Peters et al., 2013), but is there any way to mitigate the risks? Even if we cannot prevent the sea levels from rising or storms raging, maybe we can protect our coastal ecosystems and cities by restoring resilience in social-ecological systems with ecosystem based defence strategies.
Concept of resilience
Resilience was first introduced as an ecological concept by Holling in 1973, the idea mainly referring to dynamic ecosystems that can persist in the face of disturbances. High ecological resilience is closely linked to high biodiversity of ecosystems (e.g. Oliver et al., 2015; Worm et al., 2006). As people are increasingly seen as an integral part of the biophysical world (Egan, Hjerpe & Abrams, 2011), our current understanding of resilience now also includes the human dimension. According to one definition, resilience is the capacity of social-ecological system to sustain a desired set of ecosystem services in the face of disturbance and ongoing evolution and change (Biggs et al., 2012, p. 423).
From human-engineered to ecosystem based defences
For a long time, coastal hazard prevention relied solely on so called “hard solutions”, such as building of sea walls and dykes (Slobbe et al., 2013). Recently there has been a shift towards “softer” approaches. These so called ecosystem-based adaptation or defence strategies aim to conserve or restore naturally resilient coastal ecosystems, such as marshes and mangroves, in order to protect human population from natural hazards (Temmerman et al., 2013). Restoring shores for protection is not a new idea, but it has gained momentum in recent years. Many volunteer groups are focused on restoring coastal ecosystems, such as the Dune Restoration Trust in New Zealand. Globally, the influential Nature Conservancy funds a project called Coastal Resilience, which aims to reduce coastal risks to communities with nature-based solutions (Coastal Resilience, 2016).
Restoring dune vegetation can help reduce erosion, while increasing and maintaining the resilience of coastal zones (Silva, Martínez, Odériz, Mendoza, & Feagin, 2016). Coastal ecosystems, for example forested wetlands and marshes, can play a significant role in reducing the influence of waves (Fig. 1) and floods (Danielsen et al., 2005; Hey & Philippi, 1995; Mitsch & Gosselink, 2000; Seabloom et al., 2013). In southeast India coastal zones with intact mangrove forests and tree shelterbelts were significantly less affected by the catastrophic Boxing Day tsunami in 2004, than the areas where coastal vegetation had been removed (Danielsen et al., 2005). Coastal vegetation can also buffer gradual phenomena such as sea-level rise or tidal changes (Feagin et al., 2009).

Figure 1. A simple figure showing how the wave impact is reduced in healthy coastal habitats due to the buffering effect of different coastal ecosystems, such as marshes. The Nature Conservancy.
One of the benefits of ecosystem-based strategies compared to traditional human-engineered solutions is that they are more cost-efficient. For example, investment of US$1.1 million on mangrove restoration to protect rice fields in coastal Vietnam has been estimated to save US$7.3 million per year in dyke maintenance (Reid & Huq, 2005). In addition, almost 8,000 local families have been able to improve their livelihoods and thus their resilience by harvesting marine products in the replanted mangrove areas (Reid & Huq, 2005).
It has been argued that healthy natural ecosystems are more effective than man-made structures in coastal protection (Costanza, Mitsch, & Day, 2006). For example, the devastating effects of the 2005 flood in New Orleans could partially have been avoided, if the wetlands surrounding the city had not been modified by humans, thus preventing the delta system absorbing changes in water flows (Costanza et al., 2006). The problem is, due to anthropogenic stressors, not many coastal habitats are healthy or in a natural state. This is something that restoration aims to change, but to really make a difference, we have a long road ahead.
Future
Significant mitigation of greenhouse gas emissions is the most crucial action that can be taken to reduce the effects of climate change, but we also need to adapt to the predicted changes by increasing ecosystem management methods sensitive to resilience (Tompkins & Adger, 2004). Traditionally, ecological restoration is based on the idea that we want to return something to its former condition. But ecosystems are not stable or static, never have been, and never will be (Willis & Birks, 2006). The increased risk of climate change induced coastal hazards possesses a major challenge to New Zealand economically, socially and environmentally. We have approximately 18,200 kilometres of shoreline, and one of the highest coast to land area ratios in the world. Most of New Zealand’s towns and cities, including our capital city Wellington, are located by the sea. In order to survive, we need to embrace ecosystem-based solutions and aim to restore for resilience.
References
Biggs, R., Schluter, M., Biggs, D., Bohensky, E. L., BurnSilver, S., Cundill, G., . . . West, P. C. (2012). Toward Principles for Enhancing the Resilience of Ecosystem Services. In A. Gadgil & D. M. Liverman (Eds.), Annual Review of Environment and Resources, Vol 37 (Vol. 37, pp. 421-+). Palo Alto: Annual Reviews
Coastal resilience. (2016). http://coastalresilience.org/ [Accessed on 22 April 2016].
Costanza, R., Mitsch, W. J., & Day, J. W. (2006). A new vision for New Orleans and the Mississippi delta: applying ecological economics and ecological engineering. Frontiers in Ecology and the Environment, 4(9), 465-472. doi:10.1890/1540-9295(2006)4[465:ANVFNO]2.0.CO;2
Danielsen, F., Sørensen, M. K., Olwig, M. F., Selvam, V., Parish, F., Burgess, N. D., . . . Suryadiputra, N. (2005). The Asian Tsunami: A Protective Role for Coastal Vegetation. Science, 310(5748), 643-643. Retrieved from http://science.sciencemag.org/content/310/5748/643.abstract
Egan, D., Hjerpe, E. E., & Abrams, J. (2011). Why people matter in ecological restoration. In Human Dimensions of Ecological Restoration (pp. 1-19). Island Press/Center for Resource Economics
Emanuel, K. (2005). Increasing destructiveness of tropical cyclones over the past 30 years. Nature, 436(7051), 686-688. doi:http://www.nature.com/nature/journal/v436/n7051/suppinfo/nature03906_S1.html
Feagin, R. A., Lozada-Bernard, S. M., Ravens, T. M., Möller, I., Yeager, K. M., & Baird, A. H. (2009). Does vegetation prevent wave erosion of salt marsh edges? Proceedings of the National Academy of Sciences, 106(25), 10109-10113.
Gornitz, V. (1991). Global coastal hazards from future sea level rise. Palaeogeography, Palaeoclimatology. Palaeoecology 89(4): 379–398
Hey, D. L., & Philippi, N. S. (1995). Flood Reduction through Wetland Restoration: The Upper Mississippi River Basin as a Case History. Restoration Ecology, 3(1), 4-17. doi:10.1111/j.1526-100X.1995.tb00070.x
Holling, C. S. (1973). Resilience and Stability of Ecological Systems. Annual Review of Ecology and Systematics, 4, 1-23. Retrieved from http://www.jstor.org.helicon.vuw.ac.nz/stable/2096802
Martínez, M. L., Intralawan, A., Vázquez, G., Pérez-Maqueo, O., Sutton, P., & Landgrave, R. (2007). The coasts of our world: Ecological, economic and social importance. Ecological Economics, 63(2–3), 254-272. doi:http://dx.doi.org/10.1016/j.ecolecon.2006.10.022
McGranahan, G., Balk, D., & Anderson, B. (2007). The rising tide: assessing the risks of climate change and human settlements in low elevation coastal zones. Environment and urbanization, 19(1), 17-37.
Millennium Ecosystem Assessment. (2005). Ecosystems and Human Well-being: Biodiversity Synthesis. World Resources Institute, Washington, DC
Mitsch, W. J., & Gosselink, J. G. (2000). The value of wetlands: importance of scale and landscape setting. Ecological Economics, 35(1), 25-33. doi:http://dx.doi.org/10.1016/S0921-8009(00)00165-8
Nicholls, R. J., & Lowe, J. A. (2004). Benefits of mitigation of climate change for coastal areas. Global Environmental Change, 14(3), 229-244. doi:http://dx.doi.org/10.1016/j.gloenvcha.2004.04.005
Nicholls, R.J., P.P. Wong, V.R. Burkett, J.O. Codignotto, J.E. Hay, R.F. McLean, S. Ragoonaden and C.D. Woodroffe. (2007). Coastal systems and low-lying areas. Climate Change 2007: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, M.L. Parry, O.F. Canziani, J.P. Palutikof, P.J. van der Linden and C.E. Hanson, Eds., Cambridge University Press, Cambridge, UK, 315-356.
Oliver, T. H., Heard, M. S., Isaac, N. J., Roy, D. B., Procter, D., Eigenbrod, F., . . . Petchey, O. L. (2015). Biodiversity and resilience of ecosystem functions. Trends in Ecology & Evolution, 30(11), 673-684.
Peters, G. P., Andrew, R. M., Boden, T., Canadell, J. G., Ciais, P., Le Quere, C., . . . Wilson, C. (2013). The challenge to keep global warming below 2 [deg]C. Nature Clim. Change, 3(1), 4-6. doi:http://www.nature.com/nclimate/journal/v3/n1/abs/nclimate1783.html#supplementary-information
Reid, H., & Huq, S. (2005). Climate change-biodiversity and livelihood impacts. Tropical forests and adaptation to climate change, 57.
Seabloom, E. W., Ruggiero, P., Hacker, S. D., Mull, J., & Zarnetske, P. (2013). Invasive grasses, climate change, and exposure to storm-wave overtopping in coastal dune ecosystems. Global Change Biology, 19(3), 824-832. doi:10.1111/gcb.12078
Silva, R., Martínez, M. L., Odériz, I., Mendoza, E., & Feagin, R. A. (2016). Response of vegetated dune–beach systems to storm conditions. Coastal Engineering, 109, 53-62. doi:http://dx.doi.org/10.1016/j.coastaleng.2015.12.007
Slobbe, E., Vriend, H. J., Aarninkhof, S., Lulofs, K., Vries, M., & Dircke, P. (2013). Building with Nature: in search of resilient storm surge protection strategies. Natural Hazards, 66(3), 1461-1480. doi:10.1007/s11069-013-0612-3
Temmerman, S., Meire, P., Bouma, T. J., Herman, P. M. J., Ysebaert, T., & De Vriend, H. J. (2013). Ecosystem-based coastal defence in the face of global change. Nature, 504(7478), 79-83. doi:10.1038/nature12859
Tompkins, E. L., & Adger, W. N. (2004). Does Adaptive Management of Natural Resources Enhance Resilience to Climate Change? Ecology and Society, 9(2). Retrieved from http://www.ecologyandsociety.org/vol9/iss2/art10/
Willis, K. J., & Birks, H. J. B. (2006). What is natural? The need for a long-term perspective in biodiversity conservation. Science, 314(5803), 1261-1265.
Worm, B., Barbier, E.B., Beaumont, N., Duffy, J.E., Folke, C., Halpern, B.S., Jackson, J.B., Lotze, H.K., Micheli, F., Palumbi, S.R. and Sala, E., (2006). Impacts of biodiversity loss on ocean ecosystem services. Science, 314(5800), pp.787-790.
Moving Towards an Inclusive Restoration Model: The Crumbling of a Paradox
Posted: May 28, 2015 Filed under: 2015, ERES525, Research Essay Leave a commentBy Jake Tessler
Wilderness is untouched and untamed. Wilderness is romantic and remote. Wilderness exists outside of our sphere of influence, and must continue to do so in order to thrive. These sentiments are commonly held notions of nature in its purest, “wildest” form. But how do we reconcile these conceptualizations of nature with an environment that sees an increasing need for human intervention to countermand a mounting number of detrimental anthropogenic effects? This conflicting dichotomy forms the basis for what Throop and Purdom (2006) have termed the “participation paradox”. One of the key distinctions they have made is that participatory restoration tends to focus on integrating people into natural surroundings, not mitigating the impacts of human development. This, necessarily, puts participatory conservation efforts at odds with wilderness preservation. Their examination led to the conclusion that participatory conservation efforts should be isolated and limited to human-dominated landscapes to the extent possible.
However, recent emphasis on the integration of human experiences and activities into restoration and conservation projects represent an engagement-based paradigm shift, seeking to demystify “wilderness” and expand perceptions of nature (see Marris 2011). Specifically, aligning the process of restoration ecology with site-specific social and cultural importance can facilitate a bond between people and their natural surroundings. This form of participation-based ‘focal restoration’ requires that inclusive restoration practices concentrate less on the “wildness” of an area and instead emphasise the inherent social, cultural and ecological values (Higgs 2003).
Public engagement in restoration efforts has been described as critical to successfully navigate the complex and often controversy-laden landscape surrounding the politics associated with ecological restorations (Gobster 2000). Recent studies have sought to emphasize the importance and similarly controversial nature of moving public engagement out of town halls and community forums and into the field through volunteerism and citizen scientist-based project direction (Buzier et al. 2012; Theobald et al. 2015).
There are generally combinations of factors that motivate individuals towards environmental volunteering. Some of the most powerful have been found to be a sense of general environmental stewardship and location-specific attachment (Measham & Barnett 2008), as well as “atonement” for either past or present environmental damage (Clewell & Aronson 2006). Famed American biologist E.O. Wilson explores the evolutionary and philosophical aspects of such attachments in his 1984 book Biophilia. In the three decades following, a variety of interpretations and emphases on this subject have been discussed (see Nisbet et al. 2009).
Regardless of the impetus that compels an individual to volunteer time and effort, the will to do so is predicated upon a bond one feels, or desires to feel, with the natural world. However, capitalizing on the availability of volunteer labor is often met with hesitation from land managers helming restoration projects. A 2014 survey of land manager opinions towards to use of volunteer labor in their projects over the next 12 months showed that while respondents were evenly split as to their intentions, the largest block of respondents (nearly 25%) claimed to be undecided as to their inclusion of volunteer labor (Bruce et al. 2014). Commonly expressed reasons for this ambivalence included the lack of access to experienced and properly trained volunteers. This carries with it increased safety concerns as well as increased financial liabilities.
The key to surmounting these difficulties may lie in comprehensive youth engagement. Familiarizing young people with field-based, environmental restorations has the potential to not only impart valuable technical skills and help develop the natural bond required for life-long stewardship but contains intrinsic social and psychological value (Grese et al. 2000). Unfortunately, some researchers believe that we are currently witnessing the creation of an ecological generational gap. Defined by the lack of a personally developed, human-nature relationship fostered through a technology-based, recreationally insular worldview (Kareiva & Marvier 2012; Miller 2005; Pergams & Zaradic 2008), this gap has wide-ranging implications for active management restorations.
Student-based restoration projects, while usually not cost effective, have been shown to help create awareness and increase the appreciation of participants to their local surroundings (Evans et al. 2012). It is imperative to foster these lifelong human-nature relationships in young people and to do so through public education campaigns and early volunteer engagement. The continued and concerted implementation of a participatory, youth-based model should be considered paramount for the long-term success of future ecological restoration practice. Although incursions into established wilderness areas should be done so with care, we must accept that delineations which separate humans from wilderness are inherently, and subjectively, value-based. As such, it is imperative that these values are allowed not only to persevere, but evolve in order to retain relevancy.
References
Bruce MC, Newingham BA, Harris CC, Krumpe EE 2014. Opinions toward using volunteers in ecological restoration: a survey of federal land managers. Restoration Ecology 22: 5-12.
Buzier M, Kurz T, Ruthrof K 2012. Understanding restoration volunteering in a context of environmental change: in pursuit of novel ecosystems or historical analogues? Human Ecology 40: 153-160.
Clewell AF, Aronson J 2006. Motivations for the restorations of ecosystems. Conservation Biology 20: 420-428.
Evans E, Ching CC, Ballard HL 2012. Volunteer guides in nature reserves: exploring environmental educators perceptions of teaching, learning, place and self. Environmental Education Research 18: 391-402.
Gobster PH 2000. Restoring nature: human actions, interactions, and reactions. In: Gobster PH, Hull B eds. Restoring nature: perspectives from the social sciences and humanities. Washington, D.C., Island Press.
Grese RE, Kaplan R, Ryan RL, Buxton R 2000. Psychological benefits of volunteering in stewardship programs. In: Gobster PH, Hull B eds. Restoring nature: perspectives from the social sciences and humanities. Washington, D.C., Island Press.
Higgs E 2003. Nature by design: people, processes, and ecological restoration. Cambridge, MIT Press.
Kareiva P, Marvier M 2012. What is Conservation Science? BioScience 62: 962-969.
Marris E 2011. Rambunctious garden: saving nature in a post-wild world. New York, Bloomsbury USA.
Measham TG, Barnett GB 2008. Environmental volunteering: motivations, modes and outcomes. Australian Geographer 39: 537-552.
Miller JR 2005. Biodiversity conservation and the extinction of experience. Trends in Ecology and Evolution 20: 430–434.
Nisbet EK, Zelenski JM, Murphy SA 2009. The Nature Relatedness Scale: Linking individuals’ connection with nature to environmental concern and behavior. Environment and Behavior 41: 715-740.
Pergams ORW, Zaradic PA 2008. Evidence for a fundamental and pervasive shift away from nature-based recreation. Proceedings of the National Academy of Sciences 105: 2295–2300.
Theobald EJ, Ettinger AK, Burgess HK, DeBey LB, Schmidt NR, Froelich HE, Wagner C, HilleRisLambers J, Tewksbury J, Harsch MA, Parrish JK 2015. Global change and local solutions: tapping the unrealized potential of citizen science for biodiversity research. Biological Conservation 181: 236-244.
Throop W, Purdom R 2006. Wilderness restoration: the paradox of public participation. Restoration Ecology 14: 493-499.